Next: Summary Up: Dialkyl Side Chain Substituents Previous: Poly(dialkylsilanes)
One class of materials with excellent potential for blue LED applications are ![]() -conjugated polymers incorporating poly(
-conjugated polymers incorporating poly(![]() -phenylene) based backbones. In this case it is the torsional twist between adjacent phenylene rings which leads to suitable increases in the band gap. PPPs can be forcibly planarized by replacing neighbor hydrogen pairs with a bridging carbon (coupled to solubilizing side chains) to give a class of ladder type polymers[104] (LPPPs). LPPPs have band gaps in the range of 2.7 eV. These rigid rod polymers tend to have relatively low molecular weights and more limited processibility than more conformationally flexible polymers.
-phenylene) based backbones. In this case it is the torsional twist between adjacent phenylene rings which leads to suitable increases in the band gap. PPPs can be forcibly planarized by replacing neighbor hydrogen pairs with a bridging carbon (coupled to solubilizing side chains) to give a class of ladder type polymers[104] (LPPPs). LPPPs have band gaps in the range of 2.7 eV. These rigid rod polymers tend to have relatively low molecular weights and more limited processibility than more conformationally flexible polymers.
Polyfluorenes (or PFOs, see Fig. 7), in which only alternating pairs of phenylene rings are fused, represent structural intermediates to PPPs and LPPPs and have a number of excellent physical and electronic properties[105,106,107]. They are known for high solid-state quantum efficiencies, good electron mobilities and exceptional thermal and chemical stability in inert environments. A large number of oligomer and polymer derivatives have been synthesized[108,109]. Of these, many recent reports address a relatively select subset of linear and branched dialkyl-substituted fluorenes. Poly(9,9-(di ![]() -alkyl)fluorenes), especially PF8 (dioctyl), PF6 (dihexyl) and PF2/6 [di(2-ethylhexyl)], have received extra attention because these polymers are mesomorphic[110,111] and, additionally, there are reports of multiple crystalline phases as well. Thus there are special opportunities for studying PFO electronic and optical properties in response to systematic molecular level structural changes. In fact this complex phase behavior extends to oligomeric forms of fluorene[109].
-alkyl)fluorenes), especially PF8 (dioctyl), PF6 (dihexyl) and PF2/6 [di(2-ethylhexyl)], have received extra attention because these polymers are mesomorphic[110,111] and, additionally, there are reports of multiple crystalline phases as well. Thus there are special opportunities for studying PFO electronic and optical properties in response to systematic molecular level structural changes. In fact this complex phase behavior extends to oligomeric forms of fluorene[109].
The branched PF2/6 polymer includes one chiral center on each alkyl side chain and so this polymer is nominally an atactic and achiral racemic mixture of (S) and (R) enantiomers. Scherf and coworker[112] have found that oriented and subsequently annealed thin films and fibers form a crystalline phase that incorporates an average chain structure consisting of either ![]() or
or ![]() helices. Ab initio quantum chemical calculations of the singlet S
helices. Ab initio quantum chemical calculations of the singlet S![]() -S
-S![]() electronic transition in fluorene dimers best correlate the PF2/6 PL (
electronic transition in fluorene dimers best correlate the PF2/6 PL (![]() 258 nm) with a 144
258 nm) with a 144![]() backbone torsion angle and a
backbone torsion angle and a ![]() helix. A full 3D model of the main chain and side chain local structure and packing has not been reported. PF2/6's which include an excess of one specific side chain enantiomer emit circularly polarized light[113] and this general effect highlights the relationship between molecular level structure and optical properties.
helix. A full 3D model of the main chain and side chain local structure and packing has not been reported. PF2/6's which include an excess of one specific side chain enantiomer emit circularly polarized light[113] and this general effect highlights the relationship between molecular level structure and optical properties.
The PF8 derivative is equally as interesting because it can be studied in two
distinct backbone conformations often referred to as the ![]() or
or ![]() phase. The
phase. The ![]() type conformation is claimed to be more planar and less disordered than the
type conformation is claimed to be more planar and less disordered than the ![]() form. Increased planarity implies a reduced interband transition energy and there is an approximate 100 mV red shift in the PL spectra of the
form. Increased planarity implies a reduced interband transition energy and there is an approximate 100 mV red shift in the PL spectra of the ![]() form in comparison films of pure
form in comparison films of pure ![]() phase. There are also additional differences in both the steady-state and time-resolved optical properties[114,115] and these will strongly impact LED behavior. As an example, Fig. 13 displays typical optical absorption and PL spectra at two reduced temperatures in PF8 thin films with different processing histories. The right panel is from a film having PFO chains residing in just the
phase. There are also additional differences in both the steady-state and time-resolved optical properties[114,115] and these will strongly impact LED behavior. As an example, Fig. 13 displays typical optical absorption and PL spectra at two reduced temperatures in PF8 thin films with different processing histories. The right panel is from a film having PFO chains residing in just the ![]() phase while the left frame is from a second sample containing a large fraction of
phase while the left frame is from a second sample containing a large fraction of ![]() type chains. Conversion to the
type chains. Conversion to the ![]() phase is facilitated by annealing samples in the presence of solvent vapor (typically toluene at temperatures near room temperature) while thermal quenching, from a conformationally disordered high temperature mesophase, to final temperatures below 260 K fully suppresses
phase is facilitated by annealing samples in the presence of solvent vapor (typically toluene at temperatures near room temperature) while thermal quenching, from a conformationally disordered high temperature mesophase, to final temperatures below 260 K fully suppresses ![]() phase formation. In both cases the distinctive Franck-Condon vibronic structure is resolved in the PL spectra and, as indicated, the series of peaks correspond to singlet exciton decay with creation of zero (0-0) one (0-1) or more (0-2 and higher order) skeletal phonons.
phase formation. In both cases the distinctive Franck-Condon vibronic structure is resolved in the PL spectra and, as indicated, the series of peaks correspond to singlet exciton decay with creation of zero (0-0) one (0-1) or more (0-2 and higher order) skeletal phonons.
The temperature dependent differences in the UV-vis absorption of each sample are relatively modest but the thermal evolution of the PL in the ![]() phase sample is drastically different. At even 250 K the vibronic overtones of the
phase sample is drastically different. At even 250 K the vibronic overtones of the ![]() phase dominate and the peaks are very much broader. Only a very slight increase in the proportion of
phase dominate and the peaks are very much broader. Only a very slight increase in the proportion of ![]() type chains is necessary for strong
type chains is necessary for strong ![]() phase emission. These sites function as local low energy traps and there is efficient energy flow from absorbing chains in the
phase emission. These sites function as local low energy traps and there is efficient energy flow from absorbing chains in the ![]() phase. At lower temperatures the
phase. At lower temperatures the ![]() conformer PL emission monotonically sharpens and red shifts. In this case it is possible to directly correlate changes in the 0-0 peak positions and peak widths and identify characteristics from recombination of thermally equilibrated singlet excitons at the bottom of an electronic band[116]. Many of these results are consistent with the hypothesis of improved structural order in the
conformer PL emission monotonically sharpens and red shifts. In this case it is possible to directly correlate changes in the 0-0 peak positions and peak widths and identify characteristics from recombination of thermally equilibrated singlet excitons at the bottom of an electronic band[116]. Many of these results are consistent with the hypothesis of improved structural order in the ![]() phase conformation.
phase conformation.
![\includegraphics[width=4.25in]{figs/pf8_plabs}](img67.png) |
New, preliminary results are shown in Fig. 14 from a thin film PF6 sample and these further document the strong sensitivity of PFO behavior to even small changes in the side chain structure (in this case, alkyl length). This figure includes selected PL spectra observed just after casting, after a low temperature anneal and, finally, after heating the sample to 200 ![]() C. The as cast and immediately cooled sample exhibits low temperature PL spectra quite comparable to those reported for the PF2/6 polymer[117]. Extended annealing at or somewhat below room temperature is associated with a gradual loss in the PL emission (not shown) and, thereafter, only modest improvements in the PL lineshape on cooling. Most notable is the development of a noticeable shoulder at short wavelengths. This characteristic remains at reduced temperatures. Although the PL features are still relatively broad at 120 K there appear to be secondary shoulders forming on the short wavelength side of the 0-0 transition (at 425 nm) suggestive of conformationally distinct emissive sites.
In studies directed towards working devices[118] similar changes to may be attributed to an irreversible process (e.g., degradation) or a more insidious process such as molecular aggregation[119]. In this instance the PF6 polymer film can be restored close to its original state simply by heating to 200
C. The as cast and immediately cooled sample exhibits low temperature PL spectra quite comparable to those reported for the PF2/6 polymer[117]. Extended annealing at or somewhat below room temperature is associated with a gradual loss in the PL emission (not shown) and, thereafter, only modest improvements in the PL lineshape on cooling. Most notable is the development of a noticeable shoulder at short wavelengths. This characteristic remains at reduced temperatures. Although the PL features are still relatively broad at 120 K there appear to be secondary shoulders forming on the short wavelength side of the 0-0 transition (at 425 nm) suggestive of conformationally distinct emissive sites.
In studies directed towards working devices[118] similar changes to may be attributed to an irreversible process (e.g., degradation) or a more insidious process such as molecular aggregation[119]. In this instance the PF6 polymer film can be restored close to its original state simply by heating to 200 ![]() C and then cooling. This evolution is nearly the antithesis of the
C and then cooling. This evolution is nearly the antithesis of the ![]() -
-![]() transition recorded in the PF8 polymer.
transition recorded in the PF8 polymer.
![\includegraphics[width=4.2in]{figs/threerows}](img68.png) |
Changes in optical and electronic properties often signify wholesale evolution in chain packing and crystal structure. Explicit knowledge of these PFO properties is still sparse but there is now ample experimental evidence for an extensive polymorphism[120]. In a series of beautiful bulk PF8 fiber X-ray diffraction (XRD) photographs, Grell et al.[121] have documented the appearance of multiple structural phases with varying degrees of molecular order. Micromorphology can also impact crystal structure and grazing incidence XRD studies[122,123] have recently shown that PFO thin films can adopt different structural forms and/orientations. Quantitative molecular level models are still unavailable.
These aforementioned X-ray results generally document structural phase behavior and polymorphism at elevated temperatures (typically above 320 K) while the PF6 and PF8 examples just discussed exhibit significant effects, in terms of optical spectroscopy, from extended thermal processing at temperatures ranging from 260 K to 320 K. The only exception is the necessity of heating to temperatures associated with thermotropic mesophase in order to effectively erase the thermal history. Recent studies now suggest that local molecular level structural relaxation of the PFO backbone can also precede crystallization and thus actually facilitate structural evolution to new crystalline forms[114,116].
Figure 15 further emphasizes the wide spread occurrence of polymorphic phase behavior in these dialkyl substituted PFOs by exhibiting selected X-ray powder diffraction profiles from a precipitated PF6 powder and the same powder after slow casting (ca. 24 h) on glass from a 1% w/w toluene solution. The precipitated polymer has a large number of relatively sharp peaks, indicating a crystalline state, superimposed on an intense monotonically decreasing small angle scattering background. The latter feature indicates large scale microscopic inhomogeneities arising from the inherently non-equilibrium nature of precipitation. Modest warming rapidly reduces the small-angle scattering background and additional heating, to just 155 ![]() C, already induces an irreversible change the X-ray pattern to a different crystalline form. (The reported PF6 thermotropic mesophase transition[110] is in excess of 160
C, already induces an irreversible change the X-ray pattern to a different crystalline form. (The reported PF6 thermotropic mesophase transition[110] is in excess of 160 ![]() C.) The lowest temperature data is qualitatively the same but the apparent merging of some peaks is suggestive of highly anisotropic thermal expansion coefficients. The initial diffraction curve from the cast film has a significantly different pattern than that of the powder and, more interestingly, continues to evolve slowly even at room temperature. Warming of this sample is first identified with an irreversible narrowing of the diffraction peaks (presumably due to improvements in the structure order) and then, at the highest temperature, initiates a transition to a new ordered phase which, for unknown reasons, closely matches the peak pattern of the original precipitated powder. Complementary characterization studies, such as differential scanning calorimetry, are not yet available. All told PF6 appears to be as structurally complex as PF8.
C.) The lowest temperature data is qualitatively the same but the apparent merging of some peaks is suggestive of highly anisotropic thermal expansion coefficients. The initial diffraction curve from the cast film has a significantly different pattern than that of the powder and, more interestingly, continues to evolve slowly even at room temperature. Warming of this sample is first identified with an irreversible narrowing of the diffraction peaks (presumably due to improvements in the structure order) and then, at the highest temperature, initiates a transition to a new ordered phase which, for unknown reasons, closely matches the peak pattern of the original precipitated powder. Complementary characterization studies, such as differential scanning calorimetry, are not yet available. All told PF6 appears to be as structurally complex as PF8.
![\includegraphics[width=4.2in]{figs/pfo_jps_xrd}](img69.png) |
A major limitation to a thorough and proper review of these PFOs and related
![]() -conjugated polymers having branched side chain substitutions is the overt lack of comprehensive molecular levels models as detailed and encompassing as those just described for the poly(dialkylsilanes). A likely starting point would once again be the nine basic low energy side chain conformations (see the previous section) dictated by hydrogen packing constraints and torsional motion about the two C-C bonds anchoring the alkyl side chains to the PFO carbon bridge. In this case the density of alkyl chains along the PFO backbone is very low so that additional alkyl-alkyl intrachain interactions from adjacent fluorene monomers would be relatively weak. In hydrocarbons the anti conformer clearly has lower energy than that of the two gauche conformations. Thus the lowest energy conformation for the side chain ``clan'' pair should be anti-anti. Single crystals of 9,9-(di
-conjugated polymers having branched side chain substitutions is the overt lack of comprehensive molecular levels models as detailed and encompassing as those just described for the poly(dialkylsilanes). A likely starting point would once again be the nine basic low energy side chain conformations (see the previous section) dictated by hydrogen packing constraints and torsional motion about the two C-C bonds anchoring the alkyl side chains to the PFO carbon bridge. In this case the density of alkyl chains along the PFO backbone is very low so that additional alkyl-alkyl intrachain interactions from adjacent fluorene monomers would be relatively weak. In hydrocarbons the anti conformer clearly has lower energy than that of the two gauche conformations. Thus the lowest energy conformation for the side chain ``clan'' pair should be anti-anti. Single crystals of 9,9-(di ![]() -octyl)fluorene monomers[124] adopt this structure. Single chain molecular modeling studies report this characteristic as well[125] although the anti-gauche conformations are fairly close in energy. In the absence of interchain interactions the main contributor to the backbone torsion angle will be, as it is in PPP, the H-H repulsions between nearest-neighbor fluorene units. In PFOs there will be an implicit asymmetry because the bridging carbon is strained and this bends the fluorene unit about the middle so that the two para-linked phenylene rings are not colinear. In addition the action of the di(alkyl) side chains will tend to inhibit cofacial contacts between neighboring chains and frustrate close packing and the layer type stacking as seen in P3ATs.
-octyl)fluorene monomers[124] adopt this structure. Single chain molecular modeling studies report this characteristic as well[125] although the anti-gauche conformations are fairly close in energy. In the absence of interchain interactions the main contributor to the backbone torsion angle will be, as it is in PPP, the H-H repulsions between nearest-neighbor fluorene units. In PFOs there will be an implicit asymmetry because the bridging carbon is strained and this bends the fluorene unit about the middle so that the two para-linked phenylene rings are not colinear. In addition the action of the di(alkyl) side chains will tend to inhibit cofacial contacts between neighboring chains and frustrate close packing and the layer type stacking as seen in P3ATs.
 |
All PFO discussion so far of the molecular level attributes has been qualitative. In one instance we are able to propose a tentative 3D structural model for the cast PF6 sample in the crystalline phase obtained after heating to 180 ![]() C. Preliminary analysis of this data gives peak positions and intensities qualitatively in agreement with a marginally triclinic lattice (
C. Preliminary analysis of this data gives peak positions and intensities qualitatively in agreement with a marginally triclinic lattice (![]() Å,
Å, ![]() Å,
Å, ![]() Å,
Å,
![]() ,
,
![]() and
and
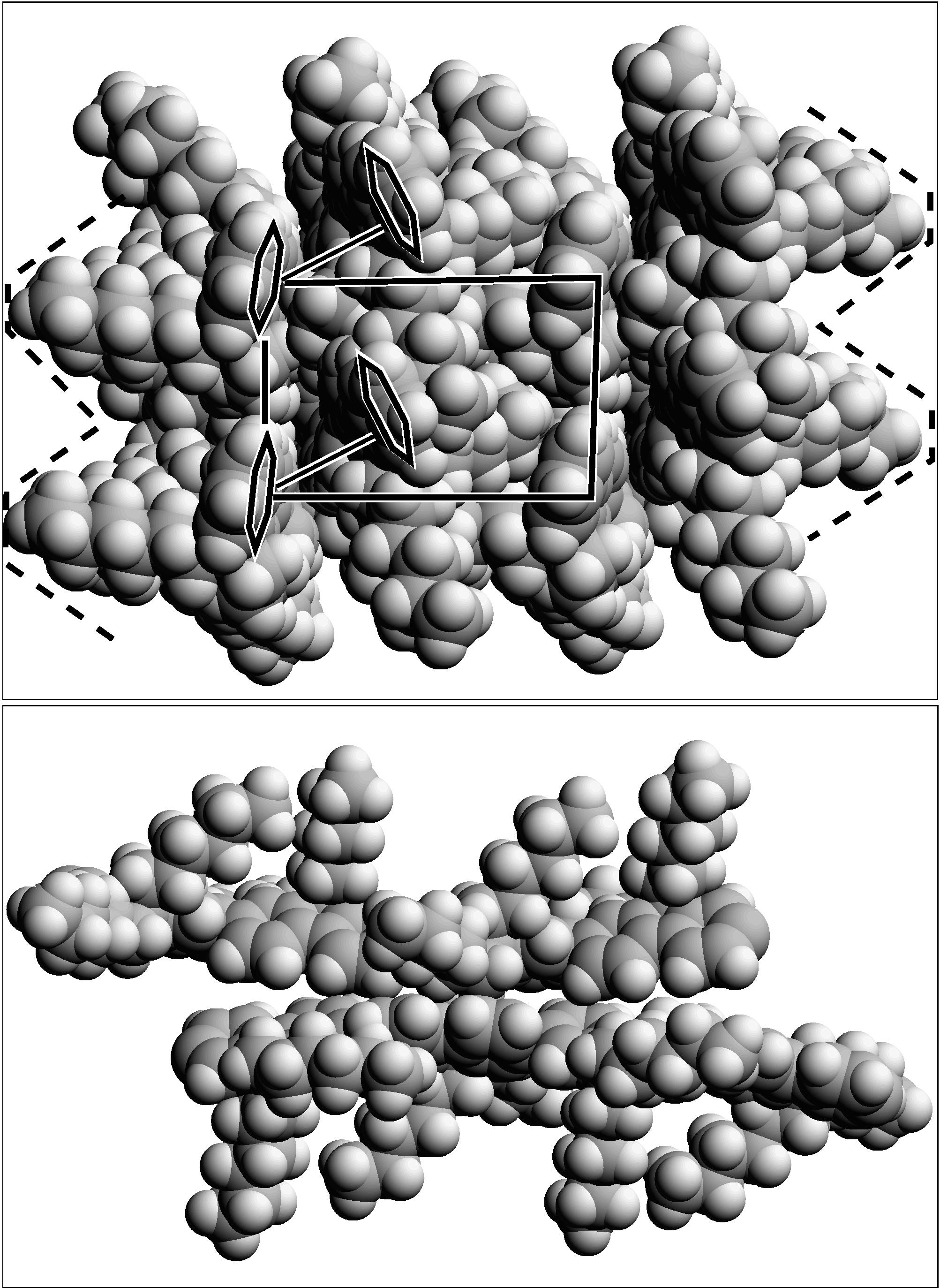
![]() ) having a two PFO chain (4 monomers per chain) unit cell and a general packing as depicted in Fig. 16. In this model the side chains adopt a sequence of anti-gauche type conformers in tandem with moderate main chain torsional twists to form a triangular shaped object (when viewed from along the
) having a two PFO chain (4 monomers per chain) unit cell and a general packing as depicted in Fig. 16. In this model the side chains adopt a sequence of anti-gauche type conformers in tandem with moderate main chain torsional twists to form a triangular shaped object (when viewed from along the ![]() -axis) on one side of the polymer strand. In this way pairs of PFO chains can achieve a partial coplanar contact with just a single neighboring chain. This cofacial type of contact will enhance transport and interchain processes only locally. This local arrangement is also viable over a large range of skeletal torsion angles. The saw-tooth side chain structure facilitates 3D ordering as well. Before this model can be considered fully self-consistent and rigorously correct more extensive molecular modeling and analysis are necessary. A more complete understanding of the molecular level structure remains a significant impediment to progress in these and many other conjugated polymers as well.
-axis) on one side of the polymer strand. In this way pairs of PFO chains can achieve a partial coplanar contact with just a single neighboring chain. This cofacial type of contact will enhance transport and interchain processes only locally. This local arrangement is also viable over a large range of skeletal torsion angles. The saw-tooth side chain structure facilitates 3D ordering as well. Before this model can be considered fully self-consistent and rigorously correct more extensive molecular modeling and analysis are necessary. A more complete understanding of the molecular level structure remains a significant impediment to progress in these and many other conjugated polymers as well.
Many other PF8 cast films [i.e., heptane, THF or even toluene] are intermediate to these two extremes and have temperature dependent properties similar to those in a recent PF8 ![]() phase study[116]. The sole PF6 sample included here (shown in Fig. 4 at left) has features more strongly identified with a high level of aggregation. In this film the position of the 0-0 300 K PL is centered near 425 nm. Hence no
phase study[116]. The sole PF6 sample included here (shown in Fig. 4 at left) has features more strongly identified with a high level of aggregation. In this film the position of the 0-0 300 K PL is centered near 425 nm. Hence no ![]() type conformations are evident. The nominal interchain excimer emission is blue shifted as well and lies centered between 515 and 520 nm. The low temperature 155 K PL spectra, which often sharpens the PL peaks, is again most consistent with the presence of a single average backbone conformation. Cooling yields only monotonic increases in the proportion of FC type emission.
type conformations are evident. The nominal interchain excimer emission is blue shifted as well and lies centered between 515 and 520 nm. The low temperature 155 K PL spectra, which often sharpens the PL peaks, is again most consistent with the presence of a single average backbone conformation. Cooling yields only monotonic increases in the proportion of FC type emission.
Winokur 2004-01-28